This truly multi-disciplinary study with experts in immunology, bioinformatics, pathology and ILC allowed us to show that there were differences in immune infiltration between ER+ ILC and ER+ NST. While majority of ILC were “cold”, some were infiltrated with immune cells, dominated by M2-like macrophages. Spatial analyses identified neighborhoods with different prognosis in ILC vs NST.
In a collaboration with the NSABP, we studied many clinico-pathological features of a large series of mixed ductal lobular cancer, and concluded that mixed IDC/ILC were more similar to ILC than IDC/NST. It is important to note that the WHO definition of mixed IDC/ILC has recently changed – the proportion of the special subtype in mixed tumors has decreased from 50% to 10%. It will be challenging to apply conclusions from our study to mixed cases with different definitions. These changes not only reflect the complicated nature of the mixed IDC/ILC tumors, but also the ever-changing definition. It is likely that the use of AI will help to clarify different ILC subtypes, including the mixed IDC/ILC cases.
A collaborative study between three founding members of the Great Lakes Breast Cancer Consortium – UPMC, Cleveland Clinic, and The James – identified meaningful differences in clinicopathological features between ILC and IDC/NST, analyzing data from more than 30,000 breast cancer patients. For patients with ILC we noted more lymph node involvement, diagnoses at later stage, higher grade and larger size tumors (with an increase over time), more mastectomies, and worse outcome – despite fewer cases with high recurrence scores.
Leigh Pate and Dr. Steffi Oesterreich had become close friends, and communicated often on how to partner researchers, clinicians and patient advocates. Together with other researchers and patient advocates they wrote a commentary putting forward a clear roadmap on how to make use of the unprecedented opportunity to drive forward multidisciplinary, multicenter and international collaborative research into ILC.
Using natural language processing, we screened more than 2 million de-identified patients records at UPMC in order to identify rare metastases to the eye. We identified 28 patients with orbital metastases, with a significant enrichment in patients with ILC. There was a trend that these metastases were more frequently bilateral, but less frequently co-occurring with metastases to the brain in ILC compared to NST, suggesting different routes of spread but larger studies combining data from multiple centers are necessary to confirm these findings.
We have had a number of productive collaborations with pharmaceutical companies, including Foundation Medicine. This study was the largest analysis of metastatic ILC at that point in time, and showed that mILC had an enrichment of NF1 mutations, and higher TMB (with APOBEC signature) compared to NST. Assessing sites of metastatic specimens showed fewer metastases in the liver and enrichment in reproductive sites, GI tract, omentum and bone marrow.
Limited availability of research models was a substantial barrier that at least partially accounted for the lack of research on ILC. Dr. Nilgun Tasdemir, a postdoc in the lab, performed a very elegant and thorough characterization of ILC cell lines – the start of our commitment to generate more ILC models faithfully representing different aspects of the disease. This project, with bioinformatics support from George Tseng, characterized 2D and 3D growth phenotypes of ILC cell lines. It emphasized how the unique morphology and cell–cell adhesion defects influence drug response and invasion, building a phenotypic framework for preclinical modeling.
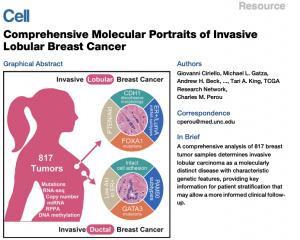
Dr. Oesterreich was honored and excited to join The Cancer Genome Atlas Network (TCGA) working group performing the largest to date comprehensive molecular analysis of ILC. This landmark paper provided a comprehensive molecular portrait of ILC, highlighting distinct genomic and transcriptomic features compared to invasive ductal carcinoma (IDC). Dr. Oesterreich contributed key expertise in lobular biology, ensuring that ILC was well represented within TCGA analyses, reflecting a major collaborative effort among over 50 institutions. The study showed that 95% of ILC cases had alterations in CDH1, that there were differences in mutations frequency (for example in PTEN, AKT, FOXA1 and TBX3), and that there are molecular variants within ILC. The conclusion was that “ILC is a clinically and molecularly distinct disease”.
This foundational study established that invasive lobular carcinoma (ILC) cell lines exhibit unique estrogen-mediated transcriptional programs and distinct tamoxifen responses compared to ductal models. Led by Dr. Matthew Sikora during his postdoctoral training in the Oesterreich–Lee laboratory, the work included collaborations with multiple Pitt faculty and collaborators such as Dr. Alana Welm (Utah), providing the first systematic evidence for differential estrogen signaling in ILC. Dr. Sikora is now Associate Professor at UC and is continuing work on ILC.